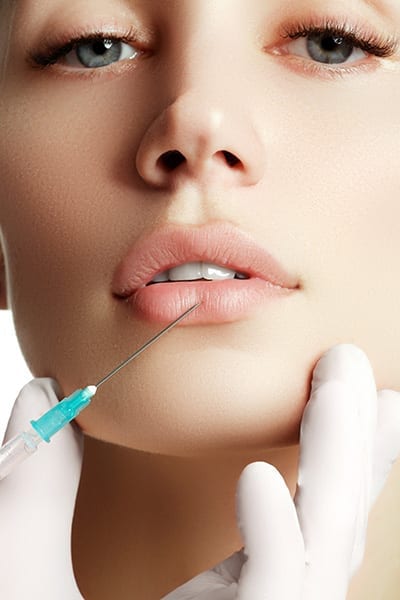
Facial Fillers
Instant Enhancement, Natural Beauty
Facial fillers are a revolutionary way to rejuvenate and enhance your appearance—without the need for surgery. At Centers for Health Promotion, we proudly offer the most trusted and advanced filler brands, including Restylane®, Sculptra®, and Aquamid®, to help you achieve your aesthetic goals with precision and care.
These cutting-edge treatments provide immediate, natural-looking results, softening wrinkles, restoring volume, and enhancing facial contours. Whether you’re looking to smooth fine lines, sculpt your features, or revitalize your complexion, facial fillers at CHP deliver stunning transformations with minimal downtime.
Facial Fillers: In-Depth Overview
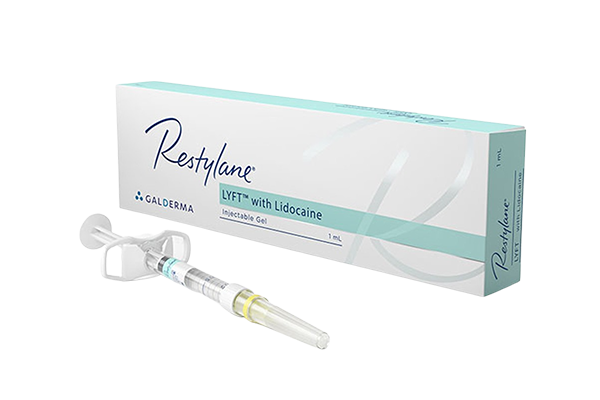
Restylane®: Temporary Filler
Restylane® is a hyaluronic acid-based (H.A.) dermal filler designed to restore volume and soften wrinkles. It’s commonly used to treat laugh lines (nasolabial folds) that extend from the nose to the corners of the mouth, as well as to enhance lip volume for a fuller, healthier, and more youthful appearance.
One of the key benefits of hyaluronic acid fillers like Restylane® is their safety profile—they carry no risk of allergic reactions, making them a popular choice for first-time filler patients.
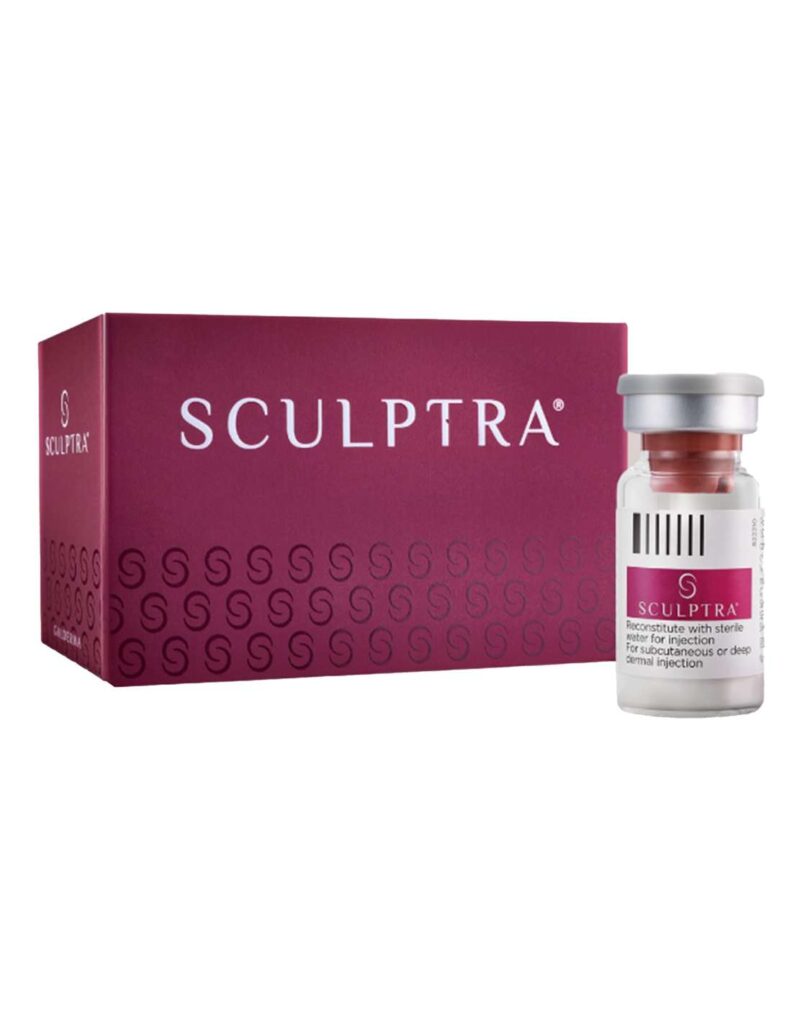
Sculptra®: Semi-Permanent Filler
Sculptra® is an excellent option for patients seeking longer-lasting results. This semi-permanent filler gradually stimulates collagen production, improving skin elasticity and volume over time. While the results take longer to become noticeable compared to temporary fillers, they offer a more durable solution for facial rejuvenation.
Sculptra® is ideal for patients who have experience with temporary fillers and are ready to transition to a treatment that delivers extended results. However, it’s important to note that semi-permanent fillers may come with slightly increased side effect risks.
Aquamid®: Permanent Filler
For those seeking a long-term solution, Aquamid® provides permanent results. This non-absorbable, non-biodegradable filler is perfect for enhancing lip volume or addressing deep nasolabial folds. Its non-allergenic properties make it a safe choice for patients looking to achieve lasting improvements without the need for repeated treatments.
Understanding Risks of Facial Fillers
Facial fillers are generally safe and well-tolerated, particularly second- and third-generation fillers that are free from animal-based extracts, eliminating the risk of allergic reactions. However, certain patients may not be candidates for fillers, including those with:
- Recurrent or active cold sores (herpes simplex)
- Insulin-dependent diabetes (Type 1)
- Pregnancy or certain systemic conditions
- Use of anti-coagulant medications
During your consultation, our experts will assess your suitability and guide you toward the best treatment options for your needs.
Facial Filler Facts
- Hyaluronic acid-based fillers were among the top non-surgical cosmetic procedures in the U.S. in 2004, with nearly 900,000 treatments performed.
- The majority of soft tissue filler patients in 2004 were between 35 and 50 years old.
- Autologous fat transfer procedures accounted for nearly 100,000 treatments that year.
- Combined, collagen and hyaluronic acid fillers represented 14% of all plastic surgery procedures performed in the United States in 2004.
Schedule Your Appointment
Facial fillers have evolved to offer a range of customizable solutions for every aesthetic goal, from temporary enhancements to permanent transformations. At Centers for Health Promotion, we’re here to help you achieve a natural, youthful appearance that fits your unique needs and lifestyle.
Schedule your consultation today to explore how facial fillers can rejuvenate and enhance your look with minimal downtime and maximum confidence.